Current Research Areas
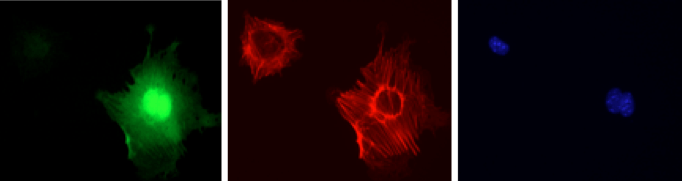
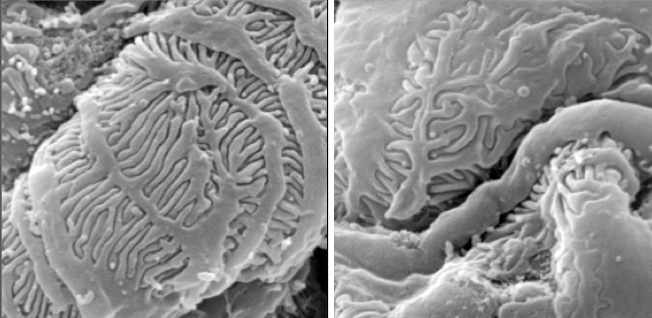
Signalling Pathways in Kidney Podocytes
Podocytes are a unique form of epithelial cells found in the kidney. They form the outer layer of the glomerulus, a specialized tuft of blood vessels responsible for blood filtration. Podocytes elaborate a series of actin-rich foot processes which give them a unique octopus-like appearance, and these foot processes are interconnected by a signalling complex known as the slit diaphragm. When the slit diaphragm is disrupted and the architecture of these cells is altered, it can lead to kidney failure. We have recently determined that the actin-associated adaptor protein Nck is required throughout life to maintain podocyte structure. Nck is recruited into the podocyte slit diaphragm through phosphotyrosine-based interactions with the scaffolding protein nephrin. We are using several complementary approaches to explore the physiological significance of nephrin/Nck signalling at the slit diaphragm, and its connection to the underlying actin cytoskeleton. We are also investigating novel signalling pathways in podocytes.
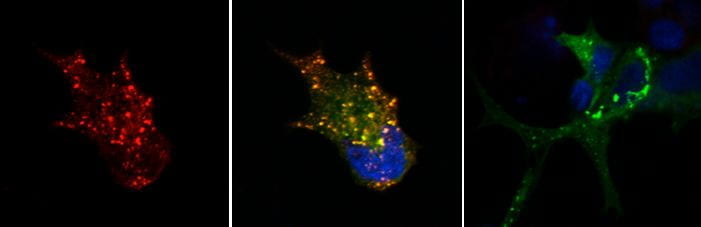
Characterization of the Novel ShcD Adaptor Protein
The Shc family of intracellular adaptor proteins function to relay phosphotyrosine-based signals from cell surface receptors to various effector proteins, and they play critical roles in cardiovascular and neural development. Although the first Shc protein was characterized in 1992, the fourth member of the family, ShcD, was not revealed until 2007. We have found that this adaptor is distinct among Shc proteins in its expression pattern and its ability to associate with binding targets, supporting the notion that elaborate diversification of Shc functions has occurred during evolution. A major objective of our lab is to characterize this newly discovered protein to determine its similarities to and differences from other Shc adaptors, and to understand its unique contributions to the complex signalling milieu of healthy and diseased cells.
Previous Projects
Cell Migration Pathways in Cardiovascular Development
Development of the cardiovascular system is dependent on angiogenesis, which serves to remodel the primitive vasculature through coordinated migration and differentiation of endothelial cells. Endothelial cell migration is regulated by intracellular signaling pathways that converge on actin to allow remodeling of the cytoskeleton, and we have found that the actin-associated adaptor protein Nck is required for this process. Using genetic approaches to selectively ablate Nck expression in the endothelium of mice, we are examining the physiological importance of Nck in cardiovascular development. Our findings are significant as alterations in the signaling pathways of cells that mediate vascular morphogenesis can lead to congenital cardiac and vessel defects, and they may also contribute to the progression of diseases such as stroke and cancer.